For products packaged in vials, after the lyophilization (freeze-drying) process is completed, a full stoppering process is generally required to ensure that the freeze-dried product is well-protected. The full stoppering process is illustrated in Figure 1. Stoppering is typically performed in a vacuum environment, though some products are stoppered after being filled with nitrogen or other protective gases, also under conditions below atmospheric pressure, to ensure that the interior of the lyophilized vial remains in a vacuum state after stoppering. Subsequently, the vials are capped after being removed from the equipment. It can be said that the stoppering and capping processes after freeze-drying are critical to ensuring that the lyophilized product remains in a sterile, isolated, and protected state.
If the stoppering quality after freeze-drying is poor, it significantly affects the product’s sterility, and the subsequent capping process cannot proceed, preventing the freeze-dried product from entering the market. This results in substantial losses for manufacturers, especially for high-value products. For instance, a single vial of medication may be worth thousands of yuan, and even if only a few dozen vials in a batch have poor stoppering, it could lead to losses amounting to tens of thousands of yuan. Therefore, improving the stability of the stoppering process and ensuring that stoppers are fully pressed into place after freeze-drying to minimize unnecessary losses is of utmost importance.
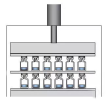
Sometimes, an entire batch may experience incomplete stoppering, or it may occur in specific localized areas. In some batches, a series of vials may also exhibit incomplete stoppering.
Vials with incomplete stoppering are directly discarded and cannot proceed to the capping process.


In Figure 3, the stopper sticking issue is quite severe, with both stoppers and vials adhering to the underside of the shelf. After stoppering, there may be only a few vials sticking to the shelf, which might go unnoticed during unloading. If these vials are crushed after unloading or fall and remain at the bottom of the chamber, residual vials may accumulate at the chamber bottom after each unloading. If these residuals enter the drainage valve and are not expelled, it can prevent the valve from fully closing, significantly affecting the vacuum level during freeze-drying.
Analysis of Causes of Freeze-Dry Stoppering Issues
Freeze-dry stoppering issues are the result of a complex interplay of multiple factors. These include the mechanical structure of the stoppering system in the freeze-dryer, the hydraulic stoppering force, the cleaning and siliconization of the stoppers, the roughness of the shelf’s underside, and the stoppering process itself.
Even if the hydraulic stoppering force is sufficient, uneven force transmission due to the shelf suspension system or localized uneven pressure can still result in incomplete stoppering. Uneven force is primarily related to the flatness of the shelf, its mechanical structure, and height variations in the vials and stoppers.
In a rod-supported shelf structure, as shown in Figure 4, the upper bracket and temperature compensation plate are integrated into a single unit. To ensure uniform force distribution, the hydraulic cylinder must be vertically aligned, with a verticality tolerance of within 0.5 mm across its full stroke. Additionally, the upper bracket must remain level when fully raised, and the vertical alignment of the cylinder is critical.
For the upper bracket, it should remain in a natural state when the shelf is fully lowered, and it must also be level. For the lower bracket, it is crucial that its upper surface remains level when naturally placed at the bottom of the chamber, while its lower surface fits tightly against the chamber. Due to the slope, the force-bearing parts of the lower bracket must be in close contact with the chamber bottom when unloaded, with a gap of less than 0.2 mm, as shown in Figure 5. This ensures that the stoppering force is fully transmitted to the bottom, guaranteeing that all stoppers are pressed into place.

Height measurements of vials and stoppers reveal that antibiotic vials typically have a height tolerance of ±0.5 mm, while stoppers have a tolerance of ±0.2 mm. The flatness of the shelf is usually controlled within 0.5 mm/m. If all dimensions deviate downward, the cumulative height difference can reach 1.2 mm. For vials and stoppers with such significant height deficiencies, the stoppering force will be noticeably insufficient, preventing the stopper from being fully pressed in. Controlling the height of vials and stoppers to keep cumulative deviations within ±0.2 mm generally ensures proper stoppering. Beyond this range, incomplete stoppering or breakage may occur.
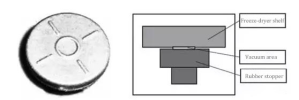
When stoppering is performed in a vacuum, the strong pressure from the shelf creates a sealed chamber inside the stopper’s ring. Upon returning to atmospheric pressure and lifting the shelf, the vacuum in this sealed chamber causes the stopper and vial to adhere to the shelf. Stoppers with a sealed ring are more prone to this issue, depending on the depth and size of the ring, making sticking more likely. Figure 7 illustrates the vacuum state formed by the stopper’s ring and the sealed chamber.
Using stoppers with an open ring at the top prevents the formation of a vacuum-sealed chamber and thus avoids sticking, as shown in Figure 8. Freeze-dry stoppers typically use open-ring designs. With an open ring in the middle, no sealed space forms after stoppering, and the vacuum is quickly released, preventing the stopper from adhering to the shelf’s underside and eliminating sticking issues.

Stopper sticking is also related to stoppering duration—the longer the duration, the more likely sticking occurs. Thus, the shortest possible duration should be used while ensuring proper stoppering. Adjusting the roughness of the shelf’s underside can help; the upper surface roughness should be Ra ≤ 0.4 μm, and the lower surface roughness should be Ra ≤ 0.8 μm, which aids in reducing sticking. In terms of process control, a reciprocating motion can be employed to mitigate the risk of stuck stoppers being lifted by the shelf. Additionally, for stopper cleaning and siliconization, validated methods should be used to obtain effective siliconization data, ensuring that the appropriate amount of silicone oil does not contribute to sticking issues.
For partial-load stoppering, at least one shelf’s worth of vials should be evenly distributed across the shelf. If there are too few vials to fill one shelf, stoppering tests should not be conducted, as this can easily crush the vials.
During full-load stoppering, it is normal to find a few vials crushed, primarily due to significant cumulative height errors from the vials, stoppers, and shelf flatness. When the height exceeds the average by 0.5 mm, the vial experiences force first and is easily crushed.
If the stoppering quality after freeze-drying is poor, it significantly affects the product’s sterility, and the subsequent capping process cannot proceed, preventing the freeze-dried product from entering the market. This results in substantial losses for manufacturers, especially for high-value products. For instance, a single vial of medication may be worth thousands of yuan, and even if only a few dozen vials in a batch have poor stoppering, it could lead to losses amounting to tens of thousands of yuan. Therefore, improving the stability of the stoppering process and ensuring that stoppers are fully pressed into place after freeze-drying to minimize unnecessary losses is of utmost importance.

Common Issues with Freeze-Dry Stoppering
The most common problems with freeze-dry stoppering include: incomplete stoppering (stopper not fully pressed in), stopper sticking after stoppering, stoppers adhering to the underside of the shelf, and vials breaking after stoppering. Among these, incomplete stoppering and stopper sticking are more frequent and are common issues encountered with many antibiotic lyophilized powder injections.Issue of Incomplete Stoppering
After stoppering, the stopper does not fully enter the vial’s mouth, as shown in Figure 2. This leaves the top of the vial unsealed, allowing external air to easily enter the vial, which prevents the subsequent capping process and significantly increases the risk of contamination. In Figure 2, it can be seen that 13 vials have incomplete stoppering. These were randomly selected as defective from nearly 3,000 stoppered vials during a stoppering test.Sometimes, an entire batch may experience incomplete stoppering, or it may occur in specific localized areas. In some batches, a series of vials may also exhibit incomplete stoppering.
Vials with incomplete stoppering are directly discarded and cannot proceed to the capping process.

Issue of Stopper Sticking After Stoppering
The problem of stopper sticking after stoppering is very common, as shown in Figure 3. Stopper sticking occurs when, after stoppering and lifting the shelf, the stopper adheres to the underside of the shelf. In some cases, only the stopper sticks, while in others, both the stopper and the vial stick to the underside of the shelf. This creates inconvenience during unloading, as it can easily cause vials to topple. Removing the stoppers and vials stuck to the shelf’s underside is also troublesome. Sometimes, during unloading, stoppers or vials fall onto other properly stoppered vials, which is difficult to handle.
In Figure 3, the stopper sticking issue is quite severe, with both stoppers and vials adhering to the underside of the shelf. After stoppering, there may be only a few vials sticking to the shelf, which might go unnoticed during unloading. If these vials are crushed after unloading or fall and remain at the bottom of the chamber, residual vials may accumulate at the chamber bottom after each unloading. If these residuals enter the drainage valve and are not expelled, it can prevent the valve from fully closing, significantly affecting the vacuum level during freeze-drying.
Issue of Vial Breakage After Stoppering
Vial breakage after stoppering occurs mainly because the vials are subjected to excessive pressure that they cannot withstand, causing them to shatter. Uneven force distribution across different areas of the vials can lead to breakage in areas experiencing higher pressure. Each vial specification has a certain pressure tolerance; when the applied pressure exceeds this limit, the vial is prone to breaking. If the actual pressure on the vial is less than its tolerance, breakage can be avoided.Analysis of Causes of Freeze-Dry Stoppering Issues
Freeze-dry stoppering issues are the result of a complex interplay of multiple factors. These include the mechanical structure of the stoppering system in the freeze-dryer, the hydraulic stoppering force, the cleaning and siliconization of the stoppers, the roughness of the shelf’s underside, and the stoppering process itself.
Analysis of Causes of Incomplete Stoppering
The root cause of incomplete stoppering is insufficient stoppering force, which fails to fully press the stopper into the vial’s mouth. The required stoppering force varies depending on the vial and stopper type. For example, a 7 mL domestic antibiotic vial requires a stoppering force of 4.9–7.84 N per stopper, while an imported 2 mL antibiotic vial requires 9.8–14.7 N per stopper. This directly affects the matching of the hydraulic stoppering force. Selecting an appropriate hydraulic system and calculating the total hydraulic pressure needed for all stoppers ensures that they are fully pressed into place.Even if the hydraulic stoppering force is sufficient, uneven force transmission due to the shelf suspension system or localized uneven pressure can still result in incomplete stoppering. Uneven force is primarily related to the flatness of the shelf, its mechanical structure, and height variations in the vials and stoppers.
In a rod-supported shelf structure, as shown in Figure 4, the upper bracket and temperature compensation plate are integrated into a single unit. To ensure uniform force distribution, the hydraulic cylinder must be vertically aligned, with a verticality tolerance of within 0.5 mm across its full stroke. Additionally, the upper bracket must remain level when fully raised, and the vertical alignment of the cylinder is critical.

For the upper bracket, it should remain in a natural state when the shelf is fully lowered, and it must also be level. For the lower bracket, it is crucial that its upper surface remains level when naturally placed at the bottom of the chamber, while its lower surface fits tightly against the chamber. Due to the slope, the force-bearing parts of the lower bracket must be in close contact with the chamber bottom when unloaded, with a gap of less than 0.2 mm, as shown in Figure 5. This ensures that the stoppering force is fully transmitted to the bottom, guaranteeing that all stoppers are pressed into place.

Height measurements of vials and stoppers reveal that antibiotic vials typically have a height tolerance of ±0.5 mm, while stoppers have a tolerance of ±0.2 mm. The flatness of the shelf is usually controlled within 0.5 mm/m. If all dimensions deviate downward, the cumulative height difference can reach 1.2 mm. For vials and stoppers with such significant height deficiencies, the stoppering force will be noticeably insufficient, preventing the stopper from being fully pressed in. Controlling the height of vials and stoppers to keep cumulative deviations within ±0.2 mm generally ensures proper stoppering. Beyond this range, incomplete stoppering or breakage may occur.
Analysis of Causes of Stopper Sticking After Stoppering
The issue of stopper sticking after stoppering is primarily related to the stopper’s shape, the stoppering process, the degree of siliconization, and the roughness of the shelf’s underside.When stoppering is performed in a vacuum, the strong pressure from the shelf creates a sealed chamber inside the stopper’s ring. Upon returning to atmospheric pressure and lifting the shelf, the vacuum in this sealed chamber causes the stopper and vial to adhere to the shelf. Stoppers with a sealed ring are more prone to this issue, depending on the depth and size of the ring, making sticking more likely. Figure 7 illustrates the vacuum state formed by the stopper’s ring and the sealed chamber.

Using stoppers with an open ring at the top prevents the formation of a vacuum-sealed chamber and thus avoids sticking, as shown in Figure 8. Freeze-dry stoppers typically use open-ring designs. With an open ring in the middle, no sealed space forms after stoppering, and the vacuum is quickly released, preventing the stopper from adhering to the shelf’s underside and eliminating sticking issues.

Stopper sticking is also related to stoppering duration—the longer the duration, the more likely sticking occurs. Thus, the shortest possible duration should be used while ensuring proper stoppering. Adjusting the roughness of the shelf’s underside can help; the upper surface roughness should be Ra ≤ 0.4 μm, and the lower surface roughness should be Ra ≤ 0.8 μm, which aids in reducing sticking. In terms of process control, a reciprocating motion can be employed to mitigate the risk of stuck stoppers being lifted by the shelf. Additionally, for stopper cleaning and siliconization, validated methods should be used to obtain effective siliconization data, ensuring that the appropriate amount of silicone oil does not contribute to sticking issues.
Analysis of Causes of Vial Breakage After Stoppering
Vial breakage after stoppering is mainly due to excessive stoppering force or individual vials experiencing pressure beyond their capacity, causing them to shatter. For full-load stoppering, the hydraulic system must be properly matched, ensuring that the hydraulic stoppering force is not excessive.For partial-load stoppering, at least one shelf’s worth of vials should be evenly distributed across the shelf. If there are too few vials to fill one shelf, stoppering tests should not be conducted, as this can easily crush the vials.
During full-load stoppering, it is normal to find a few vials crushed, primarily due to significant cumulative height errors from the vials, stoppers, and shelf flatness. When the height exceeds the average by 0.5 mm, the vial experiences force first and is easily crushed.